Is this item allowed?
Published on: 2nd December 2020 | Updated on: 18th July 2022
QUICK LINKS |
| Online Drug Tariff |
| Who Can Prescribe What? |
| Is This Prescription Form Valid? |
| Medicinal Products |
| Appliances |
Upon receipt of an NHS prescription, pharmacy staff must check if the item(s) can be prescribed on the NHS prior to dispensing. The following factors can help determine whether an item is allowed on NHS prescription:
- Nature of product – is the drug permitted on the NHS or is the appliance listed in Part IX of the Drug Tariff?
- Prescriber type – does the prescriber hold relevant registration(s) to issue prescriptions for the requested items?
- Dental or Nurse Prescribers’ formulary – are the prescribed items listed in the relevant prescribers formulary?
- Form type – is the product prescribed on a valid NHS prescription?
- Prescriber endorsements – has the prescriber annotated the prescription with the correct endorsements for e.g. ‘SLS’ for items that may only be ordered in certain circumstances?
Download our New Factsheet: Is this item allowed
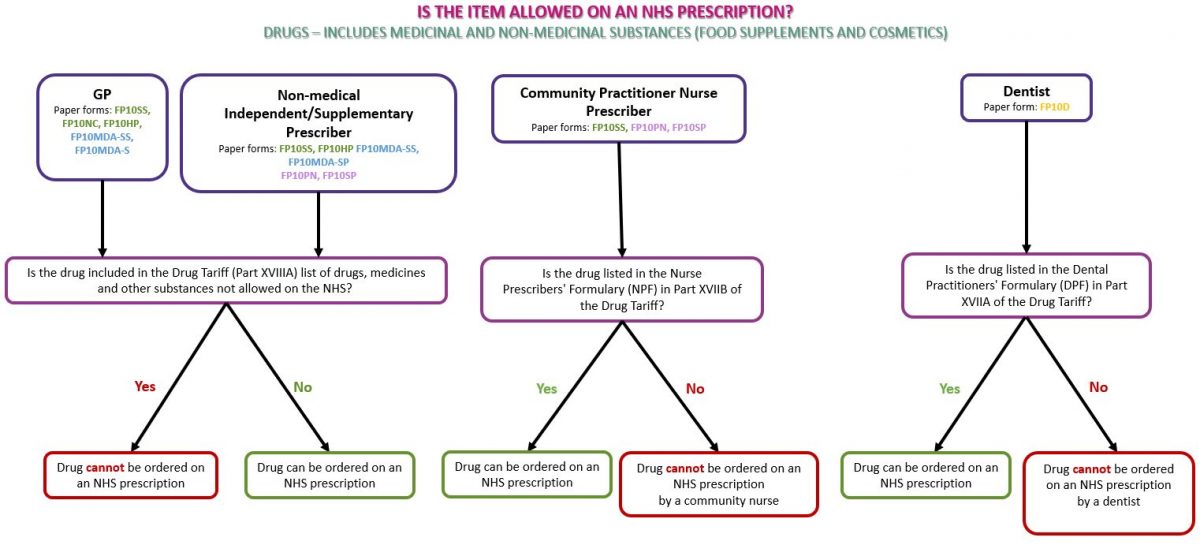
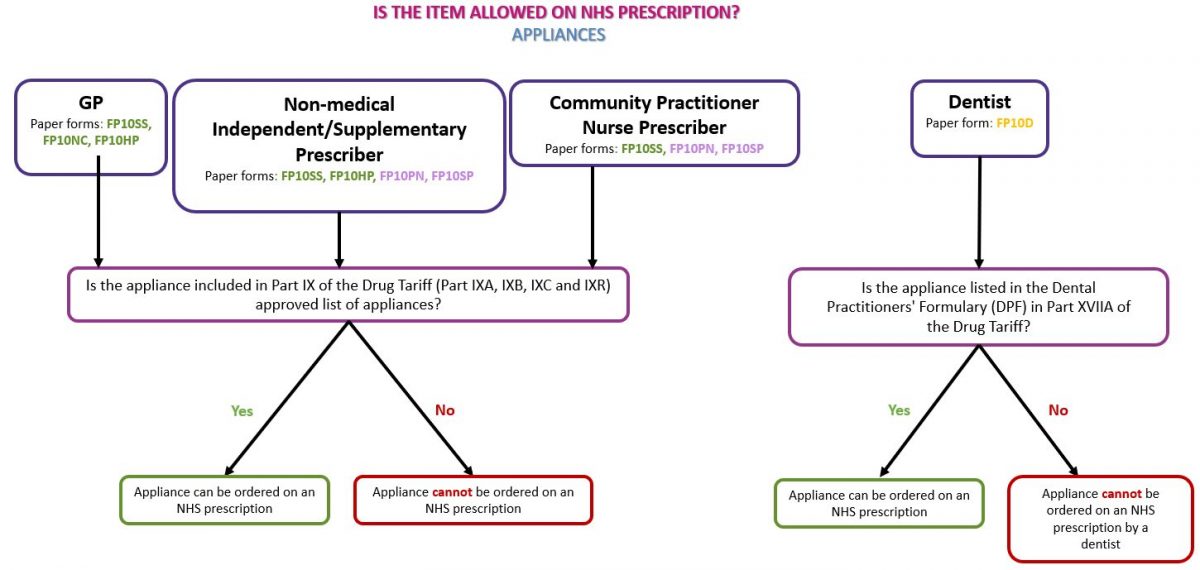
Identifying whether a product is a drug or a medical device will enable you to identify whether the item can be dispensed against an NHS prescription. Drugs Drugs refers to medical products and non-medical substances such as food supplements and cosmetics. Subject to prescribing rights (see Prescriber type below), any drug or non-medicinal substance such as food supplements or cosmetics may be prescribed on an NHS prescription if the product(s) do not appear in Part XVIIIA (drugs, medicines and other substances that may not be ordered under the NHS) of the Drug Tariff. An ‘SLS’ prescriber endorsement may be required for items that can only be ordered in certain circumstances on NHS prescriptions (see Prescriber endorsements below). See flowchart below as a guide to identifying whether the prescribed drug(s) can be dispensed on an NHS prescription: Appliances Appliances can only be prescribed on NHS prescriptions if the product is listed in Part IX (the Appliances section) of the Drug Tariff. Prescribed appliances must be an exact match for the listing in the Drug Tariff; for example the specific type and pack size must correspond with a Drug Tariff listed product. Appliances can be identified by a CE mark which indicates that the product is an appliance/medical device. The CE mark can normally be found on the packaging of a device, on the instruction leaflet and on the device itself (where practical). An appliance is allowed on NHS prescriptions as long as it is listed in Part IX of the Drug Tariff. See our page Dispensing Appliances for more information. Please note: Part IXB of the Drug Tariff states ‘Incontinence pads (including products not necessarily described as such but using the absorption principle), incontinence garments, skin wipes and occlusive devices such as female vaginal devices and male penile clamps are not prescribable under the Drug Tariff provisions.’ See flowchart below as a guide to identifying whether the prescribed appliance(s) can be dispensed on an NHS prescription: Different types of prescribers hold different prescribing rights. It is important for pharmacy staff to identify whether the prescriber holds the relevant registration(s) to issue a prescription for the requested item(s). See our page Who can prescribe what? for further information of the prescribing rights for different types of prescribers. Dental formulary Dentists can only prescribe products included in the Dental Prescribing Formulary (DPF) Part XVIIA of the Drug Tariff on an FP10D prescription. Dentists are encouraged to prescribe generically, but can prescribe by brand, any item listed in the DPF. Nurse Prescribers’ formulary Community Practitioner Nurse Prescribers who have completed the necessary training may only prescribe items appearing in the Nurse Prescribers’ Formulary for Community Practitioners list set in Part XVIIB(i) of the Drug Tariff. The following prescribing guidance is set out for Community Practitioner Nurse Prescribers in Part XVIIB(i) of the Drug Tariff: Nurses are recommended to prescribe generically, except where this would not be clinically appropriate or where there is no approved generic name. For information on the prescribing rights of nurse independent prescribers and supplementary prescribers see our page Who can prescribe what? Only certain types of prescriptions can be dispensed on the NHS, pharmacy staff must check if the prescribed product has been prescribed on a valid NHS prescription. Prescription types can be identified by the code on the bottom right of a paper prescription. Our Is this prescription form valid? page shows a table of paper prescriptions forms that can be dispensed by NHS community pharmacies and which prescribers can use them. The NHSBSA website has information on current and out of date forms for different prescriber types. SLS (The Selected List Scheme) Items included in the ‘Selected List Scheme’ which can be found in Part XVIIIB (drugs, medicines and other substances that may be ordered only in certain circumstances) of the Drug Tariff can only be prescribed for the patient groups and for the purpose(s) listed in the Tariff. Prescribers must endorse these products with ‘SLS’. If the ‘SLS’ endorsement is missing, the prescription should be returned to the prescriber for amendment to include the ‘SLS’ endorsement. Without this endorsement, the item will be returned as disallowed by the NHSBSA if it is submitted for payment by the pharmacy. Pharmacy staff cannot make the SLS endorsement themselves. CC, OC or the female symbol (♀) (Free-of-charge contraceptives) Part XVI, Clause 10 of the Drug Tariff sets out the arrangements for charging of contraceptives. Prescriptions for the following items are automatically exempt from prescription charges: However, there are some products, such as Co-cyprindiol 2000mcg/35mcg tablets, which whilst they do not appear in Part XVI, are sometimes used for contraceptive purposes. In this scenario, if the doctor has endorsed the prescription with ‘CC’, ‘OC’ or the female symbol (♀) to make it clear that the item is for contraceptive use, the patient should not incur a prescription charge. Pharmacy staff cannot apply the ‘CC’ endorsement themselves. FS (Free-of-charge items) The ‘FS’ prescriber endorsement was introduced in January 2020, to enable prescribers to indicate that a product is being prescribed for the treatment of a sexually transmitted infection (STI) and therefore can be provided to the patient free-of-charge (FOC) on an FP10 form. The ‘FS’ endorsement will operate by way of guidance; prescribers should exercise their clinical discretion in accordance with their professional duties and obligations. For sexually transmitted diseases (STDs) There is no specific list of drugs to which the ‘FS’ prescriber endorsements can be applied to. It is considered that only those conditions for which sexual contact is a main route of transmission of an infection should be ordinarily managed as “STI treatment”, for example, treatment for chlamydia would be FOC whereas treatment for candidiasis would not. Guidance on STIs that may be suitable for managing in primary care can be found in the guidelines section of the BASHH website and in the standards for the management of sexually transmitted infections. Free supply of certain COVID-19 trial medicines There is no specific list of drugs to which the ‘FS’ prescriber endorsements can be applied to. The trial medicines are usually supplied directly to eligible participants by the clinical trial team. However, because some participants in these trials will be required to take the drugs for up to 12 months and the treatments are being used outside their normal licensed indications, the Medicines and Healthcare products Regulatory Agency (MHRA) have advised that GPs should prescribe the trial medicines to patients using an FP10 prescription form or the Electronic Prescription Service (EPS). The trial medicines used in HEAL-COVID are Apixaban and Atorvastatin. STIMULATE-ICP currently uses Famotidine and Loratadine (in combination), Rivaroxaban and Colchicine. ACBS (The Borderline Substances List) In certain conditions, some foods and toiletry preparations have the characteristics of drugs. A board known as the Advisory Committee on Borderline Substances (ACBS) advises on the circumstances in which these products may be regarded as drugs. If an item which is found on the Borderline Substances list (Part XV of Drug Tariff) has not been endorsed by the prescriber with ‘ACBS’, this product can still be dispensed and will be passed for payment by NHS Prescription Services. However the prescriber may be asked by their CCG to justify why the product has been dispensed at NHS expense. Pharmacy staff are not required to endorse ‘ACBS’ on prescriptions.
The Department of Health and Social Care (DHSC) announced in March 2022 that it was extending the use of the ‘FS’ endorsement to enable free supply of certain COVID-19 trial medicines to patients participating in either the HEAL-COVID or the STIMULATE-ICP clinical trial platforms. This extended use is expected to remain in place until 31 March 2023. The DHSC took the decision to exceptionally use the ‘FS’ endorsement for this purpose as it is already available to prescribing systems and meets the requirements for this particular service.
If you are unsure whether a product is allowed, you can contact the Community Pharmacy England Dispensing and Supply Team 0203 1220 810 or info@cpe.org.uk.
Related Resources
Is This Prescription Form Valid?